AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Carbohydrates elements4/30/2023  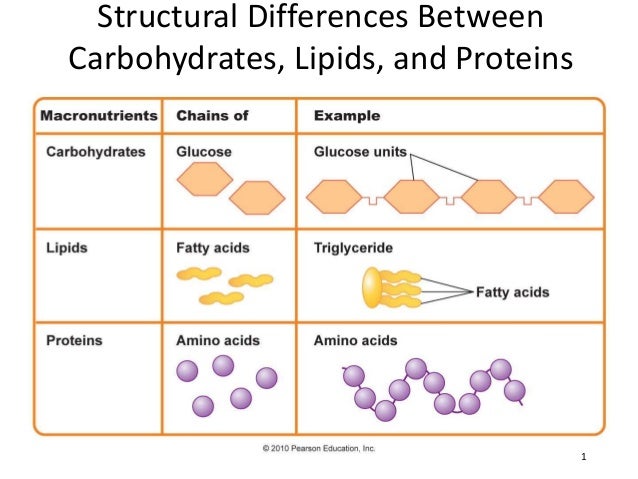
They are polyhydroxy aldehydes or ketones or form. Galactose The Glycosidic Bond Polysaccharides 1. I know this is a lot to take in, but there are several videos and articles on Khan Academy to help. Carbohydrates are composed of carbon, hydrogen, and oxygen and have a general formula that approximates CH2O. Carbohydrates are important because they hold the energy we need to power our cells. The other three are proteins, lipids and nucleic acids. 
DNA is a complete set of instructions needed for life (unless you're a virus, but that's a whole different story/debate) and RNA is used to copy DNA and to synthesize proteins. An Organic Molecule: Carbohydrates are one of four types of organic molecules, which are the molecules that make up all living organisms. RNA is single-stranded and is generally straight. Uracil links to adenine in RNA just like thymine does in DNAįinally, DNA is double-stranded and forms a double helix structure. Thymine had the chemical formula C5H6N2O2 and uracil is C4H4N2O2. Instead, it has uracil, a nucleiotide base with a slightly different chemical makeup. First, all carbohydrates are made up of the same chemical elements: carbon (that’s the carbo- part) hydrogen and oxygen, in about a two-to-one proportion, just like in H 2 O (that’s the -hydrate part) For this reason, you may see carbohydrates abbreviated as CHO in our class. You probably know that DNA has guanine, cytosine, adenine, and thymine, and that guanine links to cytosine and adenine links to thymine. PBS Schedule search for amino acid formula with fat, carbohydrate, vitamins, minerals and trace elements without phenylalanine and tyrosine, and supplemented. 
Second, while each has four nucleiotide bases, there is one difference. The common attributes of carbohydrates are that. These names describe the sugar that makes up their backbone-DNA = deoxyribose and RNA = ribose. Carbohydrates represent a broad group of substances which include the sugars, starches, gums and celluloses. To start, DNA stands for deoxyribonucleic acid, while RNA stands for ribonucleic acid. The common element which is present in both carbohydrates and proteins is. In most carbohydrates, the elements hydrogen and oxygen are present in the same 2:1 ratio as in water, thus carbo (for carbon) and hydrate (for water). Our body stores extra glucose as glycogen in muscles and liver.If you are asking what the difference between the two is, here you go. Carbohydrates, which are composed of carbon, hydrogen, and oxygen, are the major supplier of energy to the body, providing 4 kilocalories per gram. Our blood carries glucose to most cells in our body, where it is used as a source of energy. There are other types of macromolecules, but since most are organic (consisting of mostly carbon), hydrocarbons are used. These elements come together in different arrangements to form single or double units known as sugars, longer units known as starches or. So, a big chain of carbons and hydrogens is a type of macromolecule. All carbs contain carbon, hydrogen and oxygen. Hydrocarbons are just a chain of carbon atoms with hydrogens bonded to all the carbons (hence, hydro-carbon). Glycoproteins and glycolipids are often found in biological membranes, to which they are anchored by through nonpolar interactions. Carbohydrates are covalently attached to many different biomolecules, i ncluding lipids, to form glycolipids, and proteins, to form glycoproteins. \) How we use carbohydrates: we get carbohydrates in the diet as starch, glycogen, lactose, sucrose, maltose, glucose, fructose and galactose. Basically a macromolecule is a 'big molecule'. Carbohydrates in biomolecule recognition. 
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
